Ссылки tor
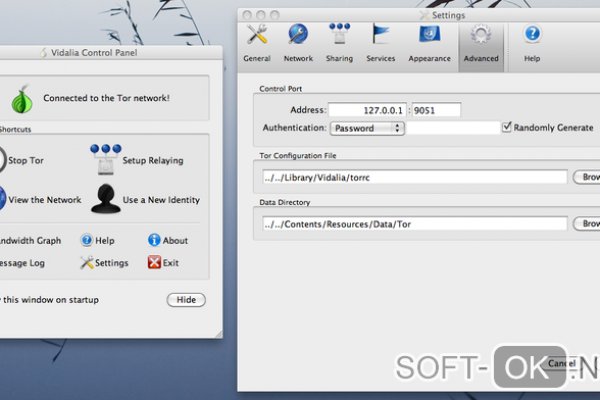
Лучшая практика по использованию TOR-браузераСуществует множество способов повысить личную безопасность при использовании в качестве браузера лукового маршрутизатора (TОR). В дополнение к использованию TOR мы рекомендуем пользоваться услугами надёжных виртуальных частных сетей (VPN) для более повышенной безопасности вашего веб-трафика. Подключение к серверу за пределами своей страны перед использованием TOR обеспечивает дополнительную защиту вашего веб-трафика от мониторинга. В браузере TOR есть ряд дополнительных настроек, которые рекомендуются в разделе “Безопасность и защита” в TOR. Во-первых, включите свой VPN. Затем,Запустите браузер TOR.Зайдите в настройки TOR и выберите “Конфиденциальность и безопасность.”Установите флажок “Удалять файлы cookie и данные сайта при закрытии браузера TOR.”Прокрутите вниз до раздела “История.” Измените настройки на “Никогда не site запоминать историю.”Прокрутите вниз до “Разрешения”: нажмите “Настройки камеры” и установите флажок “Блокировать новые запросы на доступ к вашей камере.” Сделайте то же самое относительно микрофона. Для дополнительной безопасности закройте переднюю камеру на вашем устройстве скотчем или блокировщиком.Прокрутите до раздела “Безопасность.” Нажмите “Самое безопасное.” Это отключит определенные функции в TOR, но позволит установить самое безопасное соединение.Прокрутите до пункта “Режим только для HTTPS.” Нажмите “Включить режим только для HTTPS во всех окнах.” Это гарантирует, что все ваши соединения будут зашифрованы и безопасны.Зайдите в раздел “TOR”, который расположен под разделом “Конфиденциальность и безопасность.”Установите флажок “Использовать мост.”Введите капчу.Используйте сайты https://coveryourtracks.eff.org и https://ipleak.net, чтобы определить, какая идентифицирующая информация, позволяющая однозначно идентифицировать ваш компьютер и веб-трафик, всё ещё может присутствовать.Безопасные способы подключения к TOR и “горячая линия” “ВзПП” на базе TOR:Используйте надёжный VPN-сервис, оплаченный вами. Бесплатные VPN-сервисы не всегда могут быть безопасными.Подключите своё устройство с помощью надёжного VPN-сервиса к месту за пределами страны, из которой вы получаете доступ к TOR.Запустите браузер TOR.Настройте параметры безопасности TOR по своему усмотрению.Для дополнительной безопасности используйте физический прокси-сервер, например, в кофейне или вестибюля отеля, чтобы во время соединения нельзя было определить физическое местонахождение вашего дома.Ссылки на информацию о безопасности и защите браузера TOR:Информация о мостах TOR: https://tb-manual.torproject.org/bridges/.Информация о безопасности TOR: https://tb-manual.torproject.org/security-settings/.Skip to contentМы используем файлы cookie, чтобы обеспечить максимальное удобство использования нашего веб-сайта. Если вы продолжите пользоваться сайтом, мы будем считать, что вас это устраивает.
Ссылки tor - Kraken ссылка на сайт рабочая kraken2support
Некоторые из этих луковых сайтов очень полезны, а другие просто для развлечения. Это означает, что вам не нужно будет открывать и загружать каждую ссылку из списка, чтобы убедиться, что она работает. Все сообщения хранятся зашифрованными, и администраторы Confidant не смогут получить доступ к содержимому. Он собирает данные из общедоступной сети Tor и архивирует исторические данные экосистемы Tor. На площадке ведется торговля как цифровыми, так и физическими товарами. Ни редакция, ни автор не призывают к использованию полученных знаний в практических целях и не несут ответственности за любой возможный вред, причиненный материалами данной статьи. Hidden Answers : Один из аналогов сайта Stack Overflow, где пользователи могут анонимно задать вопрос по хакингу, безопасности и приватности, криптографии, коммерции, правительстве. PrivCoin : Сервис поддерживает несколько криптовалют. Топ сайтов тор 2022 года В темной сети можно перемещаться, даже если в ней нет удобных поисковых систем. Все сайты были взяты из открытых источников и специализированных публичных каталогов. Веб-сайт доступен в сети Surface и имеет домен.onion. В даркнете другое дело: на выбор есть «Флибуста» и «Словесный Богатырь». Иначе дел с тобой вести никто не будет. Как и в случае с Гуглом, эти поисковые системы индексируют адреса.onion и, таким образом, пополняют свои базы данных. Warning Вся информация предоставлена исключительно в ознакомительных целях. Daniels Chat Daniel еще один отличный способ исследовать даркнет. Поскольку Hidden Wiki поддерживает все виды веб-сайтов, убедитесь, что вы не открываете то, что не хотите видеть. Это лучшее место для получения коротких и надежных ссылок на неизменную запись любой веб-страницы. Итак, будьте очень осторожны! На форуме действует Гарант-сервис, это обеспечит вам 100 гарантию надежности проведения сделок. ProPublica платформа журналистских расследований, которая публикует провокационные материалы на самые разные темы, от финансов до политики. Биткоин-миксеры Переводы биткоинов не являются на 100 анонимными.

О том, как зайти в даркнет я уже рассказывал в этом материале. Сегодня попробую рассмотреть какие ресурсы там есть. По вполне понятным причинам здесь не будет ссылок на сайты, занимающиеся незаконной деятельностью. Хочу подчеркнуть, это не моя личная подборка, и я не рекомендую какую-либо из представленных площадок. Это лишь список ссылок на популярные и часто упоминаемые сайты русского даркнета.Будьте внимательны! В сети орудуют мошенники.Сайты русского даркнета
doe6ypf2fcyznaq5.onion — Runion Wiki, каталог ссылок и материалы с Runion
satforumtmmmniae.onion — Подлодка — форум спутниковых пиратов. Спутниковое телевидение, оборудование для приема и декодирования. Скрытые сети и безопасность.
arhivachovtj2jrp.onion — Архива.ч — Архив имиджборд. Сохраненные треды с сайтов 2ch.hk, iichan.hk. Зеркало arhivach.org в луковой сети.
flibustahezeous3.onion — Флибуста, зеркало flibusta.net
sblib3fk2gryb46d.onion — Библиотека Траума — большое количество книг для скачивания (fb2, html, txt), без регистрации и смс.
rospravjmnxyxlu3.onion — РосПравосудие, крупнейшая картотека юристов, судей и судебных решений.
rutorc6mqdinc4cz.onion — RuTor.org, .onion зеркало торрент-трекера, скачивание без регистрации.Поисковики
3g2upl4pq6kufc4m.onion — DuckDuckGo — поисковик по клирнету. Создатели утверждают, что не собирают данные пользователей в отличии от гугла.
hss3uro2hsxfogfq.onion — not Evil.
gjobqjj7wyczbqie.onion — Candle, поисковик по сети Tor.Разумеется это далеко не полный список сайтов, поэтому если знаете ресурсы без явного криминала, то кидайте в комменты.
